Latisse Eyedrops
Looking for a proven medical solution for thin or sparse eyelashes? Frame & Focus Eye Care in Richmond, Texas offers Latisse, the only FDA-approved prescription treatment designed to increase eyelash length, thickness, and darkness. If you suffer from hypotrichosis (inadequate lash growth), our experienced eye doctors can evaluate your ocular health and prescribe this daily treatment to address the underlying issue, rather than just masking it with cosmetics.
Get Fuller Lashes—Book Your Richmond Consult!
What is hypotrichosis?
Hypotrichosis is a term used by dermatologists to describe a lack of hair growth. It’s not to be confused with alopecia, which is where previous growth has been replaced by hair loss. Hypotrichosis affects the length, number, and texture of the hair, and people who are affected develop dry, coarse, and sparse hair. The condition can affect hair anywhere on the body, including the eyelashes. As a result, patients usually have very thin, sparse eyelashes.
In many cases, hypotrichosis is hereditary, but it can also be caused by other factors, including:
- hormonal changes (such as menopause)
- inflammation of the eyelids (a condition called blepharitis)
- allergies
- overuse of eyelash curlers
If you are dissatisfied with the appearance of your eyelashes and you have been diagnosed with hypotrichosis, you may be a candidate for the highly effective, prescription-only treatment known as Latisse.
What is Latisse?
Latisse is the only FDA-approved medical treatment for eyelash hypotrichosis. This lash serum is administered in the same way as eyedrops and has been shown to increase the proportion of lashes in the growth phase of the hair cycle, meaning that they grow for longer periods of time. This helps to produce longer, fuller lashes in as little as 8 weeks.
The application of Latisse is very simple. After cleaning your eyes and ensuring all traces of cosmetics have been removed, you just apply a single drop of the solution to the base of your upper eyelashes every evening. It’s important not to use the solution on the lower eyelashes, as this could lead to excessive hair growth outside of the treatment area. In most cases, patients are recommended to apply Latisse daily for 16 weeks for maximum improvement in the thickness and length of their lashes. Results should start to become visible around 5/6 weeks after beginning the treatment and will continue to improve until you stop using Latisse.
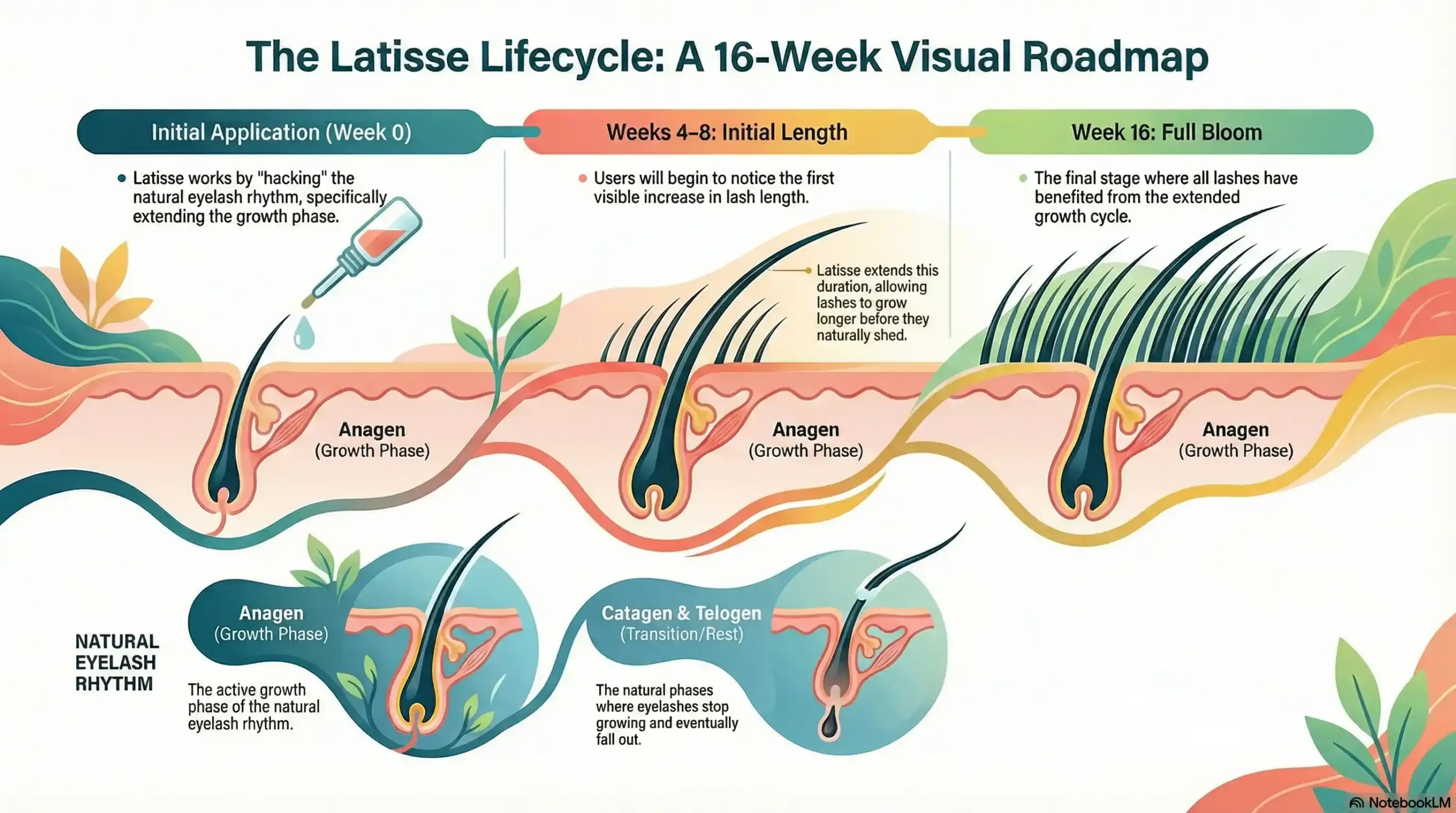
The Science of Growth: Why Latisse Needs 16 Weeks
Many patients ask why results aren’t instant. The answer lies in your biology. Your eyelashes follow a specific growth cycle, and Latisse is designed to “hack” that rhythm.
-
Anagen (Growth Phase): This is where the magic happens. Latisse extends the duration of this phase, allowing lashes to grow for a longer period before they naturally shed.
-
Catagen & Telogen (Transition/Rest): These are the phases where lashes stop growing and eventually fall out.
Because your lashes are all in different stages at any given time, you won’t see a uniform change overnight.
-
Weeks 4–8: You’ll notice initial length.
-
Week 16: This is the “Full Bloom” stage, where all lashes have benefited from the extended growth cycle.
Managing Side Effects: The “Red Eye” Solution
A common concern for new users is mild eye redness or irritation. According to Dr. Zaver, this is rarely a reaction to the medicine itself, but rather the BAK (Benzalkonium Chloride) preservative or improper application technique.
How to Stay Comfortable:
-
The “One-Drop” Rule: To avoid “flooding” the eye, use one drop of solution for both eyes. Apply to the first applicator, swipe the lid, then touch the damp applicator to the second brush.
-
Blotting is Key: Use a clean tissue to blot any excess liquid from the eyelid skin. This prevents the solution from traveling into the eye or causing skin darkening.
-
Contact Lens Care: If you wear contacts, wait at least 15 minutes after application before inserting them to prevent the preservative from absorbing into the lens.
If you struggle with chronic redness, it may be exacerbated by Dry Eye Syndrome. We recommend a comprehensive eye exam to ensure your ocular surface is healthy enough for treatment.
Latisse vs. OTC Serums: Why FDA Approval Matters
The beauty market is flooded with “lash boosters” like GrandeLASH or RapidLash. While these products are more accessible, they are classified as cosmetics, not medicine.
| Feature | Latisse (Prescription) | OTC Lash Serums |
| Active Ingredient | Bimatoprost (Proven Growth) | Peptides or Biotin (Conditioning) |
| FDA Status | FDA-Approved for Eyelash Growth | Unregulated Cosmetics |
| Results | Increased Length, Thickness & Pigment | Mostly hydration and reduced breakage |
| Safety | Monitored by an Eye Doctor | Self-monitored |
Citations and Research Sources
The information in this article is supported by current medical literature and official regulatory documentation:
1. FDA Drug Approval Documentation U.S. Food and Drug Administration. “LATISSE (bimatoprost ophthalmic solution) 0.03% – FDA Approved Drug Products.” AccessData FDA.
2. Clinical Research on Bimatoprost Efficacy Law, Simon K. “Bimatoprost in the treatment of eyelash hypotrichosis.” Clinical Ophthalmology, vol. 4, 2010, pp. 349-358. National Center for Biotechnology Information.
For more information about Latisse, or to find out if you are a good candidate for treatment, please speak to our dedicated eye care team.
FAQs
-
The active ingredient is bimatoprost. It extends the growth phase (anagen) of your natural eyelash cycle, increasing the number, length, and thickness of the lashes.




